12 February 2024
Smith+Nephew (LSE:SN, NYSE: SNN), the global medical technology company, announces the premiere of its newly acquired CARTIHEAL◊ AGILI-C◊ Cartilage Repair Implant alongside the REGENETEN◊ Bioinductive Implant at the AAOS Annual Meeting, demonstrating its leadership with products that enable biological healing for Sports Medicine. Backed by impressive clinical evidence, both technologies are transforming the way healthcare professionals treat soft tissue repair and helping improve patient outcomes versus the current standard of care.1,2
CARTIHEAL AGILI-C◊ Cartilage Repair Implant – a simple, single surgery to repair cartilage and restore bone
The CARTIHEAL AGILI-C implant is an accessible, one-step treatment for osteochondral (bone and cartilage) lesions that does not require donor tissue. It is indicated to treat a broad patient population, including traumatic, osteoarthritic and the approximately 700,0003 individuals who receive cartilage repair annually in the US. The implant has demonstrated proven superiority with twice the pain reduction1 versus the current standard of care* and has the potential to transform cartilage repair outcomes.
During its development, the CARTIHEAL AGILI-C implant received breakthrough designation status by the United States Food and Drug Administration – expedited review and approval for a product that could provide more effective treatment than existing alternatives and in the best interest of patients. It received premarket approval in 2022 and is now commercially available for sale in the US.
“I’ve already used the CARTIHEAL AGILI-C implant on a range of patients and have been very impressed with the clinical results,” said Sabrina M. Strickland, MD, orthopaedic surgeon at the Hospital for Special Surgery in New York City. “What is most exciting is that this technology represents a paradigm shift in cartilage repair and bone restoration where a lot of patients that didn’t previously have a good option can now receive treatment.”

REGENETEN Bioinductive Implant – changing the game in rotator cuff repair and expanding applications
With more than 100,000 procedures completed globally since its introduction in 2014, the REGENETEN Bioinductive Implant has had a massive impact, offering a potentially better solution for more than 1 million4 people having surgery for tendon injuries each year. The collagen-based implant supports the body’s natural healing response to facilitate the formation of new tendon-like tissue to biologically augment the existing tendon and change the course of disease progression.2,5-10
A recently completed randomized controlled trial published in December 2023, concluded that at one-year, re-tear rates for medium and large full-thickness rotator cuff tears repaired and augmented with the REGENETEN Bioinductive Implant were reduced by 68%.3

REGENETEN Bioinductive Implant for Rotator Cuff Repair
The REGENETEN Bioinductive Implant is similarly redefining healing potential for Achilles injuries when used to augment insertional or midsubstance Achilles repair. The implant creates an environment that is conducive to healing and induces tendon-like tissue growth.†,‡,5 When coupling the unique properties of the REGENETEN Bioinductive Implant with Smith+Nephew’s new procedural offerings for Achilles injuries, the solution is unmatched within the Foot and Ankle segment.
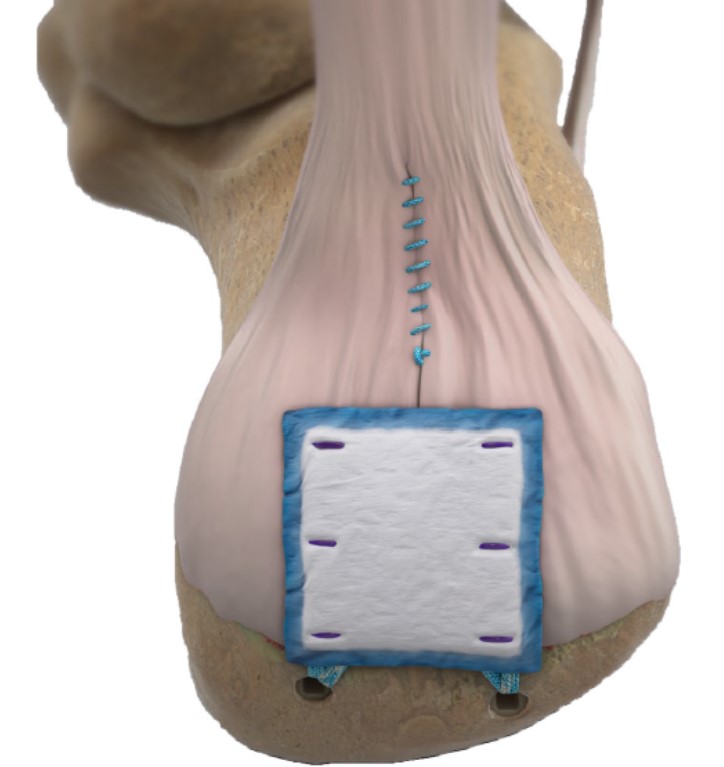
REGENETEN Bioinductive Implant for Achilles Tendon Repair
“As a leader in Sports Medicine, we are committed to bringing emerging technologies that address unmet clinical needs to our customers and their patients,” said Scott Schaffner, President Sports Medicine, Smith+Nephew. “Both REGENETEN and CARTIHEAL AGILI-C implants have the opportunity to set new benchmarks for standard of care across multiple joint repair indications and deliver better patient outcomes.”
To learn more about Smith+Nephew’s offerings that enable biological healing including the CARTIHEAL AGILI-C Cartilage Repair Implant and the REGENETEN Bioinductive Implant, please visit the Smith+Nephew booth (#5469) at the American Academy of Orthopaedic Surgeons 2024 Annual Meeting from February 12-16 in San Francisco, CA.
- ends -
Enquiries
| Media David Snyder +1 978-749-1440 Smith+Nephew | |
References
- Altschuler N, Zaslav KR, Di Matteo B, et al. Aragonite-Based Scaffold Versus Microfracture and Debridement for the Treatment of Knee Chondral and Osteochondral Lesions: Results of a Multicenter Randomized Controlled Trial. Am J Sports Med. 2023;51(4):957-967. doi:10.1177/03635465231151252
- Ruiz Ibán MA, Navlet MG, Marco SM, et al. Augmentation of a transosseous equivalent repair in posterosuperior non-acute rotator cuff tears with a bioinductive collagen implant decreases the re-tear rate at one year. A randomised controlled trial. Arthroscopy. Published online 12/27/2023.
- Medtech 360, Sports Medicine Devices, Market Analysis, US, 2018, Millennium Research Group, Inc.
- iData Research. Rotator cuff repair and reconstruction market size, share and trends analysis (2023). Available at: https://idataresearch.com/product/rotator-cuff-repair-reconstruction-market-size-share-and-trends-analysis-global-2023-2029-medsuite-includes-grafts-allografts-xenograft-synthetic-and-1-more/#. Accessed December 19, 2023.
- Bokor DJ, Sonnabend D, Deady L, et al. Evidence of healing of partial-thickness rotator cuff tears following arthroscopic augmentation with a collagen implant: a 2-year MRI follow-up. Muscles, Ligaments Tendons J. 2016;6(1):16-25.
- Schlegel TF, Abrams JS, Bushnell BD, Brock JL, Ho CP. Radiologic and clinical evaluation of a bioabsorbable collagen implant to treat partial-thickness tears: a prospective multicenter study. J Shoulder Elbow Surg. 2018 27(2):242-251.
- Van Kampen C, Arnoczky S, Parks P, et al. Tissue-engineered augmentation of a rotator cuff tendon using a reconstituted collagen scaffold: a histological evaluation in sheep. Muscles Ligaments Tendons J. 2013;3(3):229-235.
- Arnoczky SP, Bishai SK, Schofield B, et al. Histologic Evaluation of Biopsy Specimens Obtained After Rotator Cuff Repair Augmented With a Highly Porous Collagen Implant. Arthroscopy. 2017;33(2):278-283
- Bokor DJ, Sonnabend DH, Deady L, et al. Healing of partial-thickness rotator cuff tears following arthroscopic augmentation with a highly porous collagen implant: a 5-year clinical and MRI follow-up. Muscles, Ligaments Tendons J. 2019;9(3):338-347.
- McElvany MD, McGoldrick E, Gee AO, Neradilek MB, Matsen FA, 3rd. Rotator cuff repair: published evidence on factors associated with repair integrity and clinical outcome. Am J Sports Med. 2015;43(2):491-500.
* microfracture and/or debridement
† The REGENETEN Bioinductive implant is cleared for use on any tendon where there is not substantial loss of tendon tissue. REGENETEN Bone Anchors are only indicated for use in rotator cuff repair. Published clinical outcomes are for rotator cuff.
‡ As demonstrated in vivo
About Smith+Nephew
Smith+Nephew is a portfolio medical technology company focused on the repair, regeneration and replacement of soft and hard tissue. We exist to restore people’s bodies and their self-belief by using technology to take the limits off living. We call this purpose ‘Life Unlimited’. Our 19,000 employees deliver this mission every day, making a difference to patients’ lives through the excellence of our product portfolio, and the invention and application of new technologies across our three global business units of Orthopaedics, Sports Medicine & ENT and Advanced Wound Management.
Founded in Hull, UK, in 1856, we now operate in more than 100 countries, and generated annual sales of $5.2 billion in 2022. Smith+Nephew is a constituent of the FTSE100 (LSE:SN, NYSE: SNN). The terms ‘Group’ and ‘Smith+Nephew’ are used to refer to Smith & Nephew plc and its consolidated subsidiaries, unless the context requires otherwise.
For more information about Smith+Nephew, please visit www.smith-nephew.com and follow us on X, LinkedIn, Instagram or Facebook.
Forward-looking Statements
This document may contain forward-looking statements that may or may not prove accurate. For example, statements regarding expected revenue growth and trading margins, market trends and our product pipeline are forward-looking statements. Phrases such as "aim", "plan", "intend", "anticipate", "well-placed", "believe", "estimate", "expect", "target", "consider" and similar expressions are generally intended to identify forward-looking statements. Forward-looking statements involve known and unknown risks, uncertainties and other important factors that could cause actual results to differ materially from what is expressed or implied by the statements. For Smith+Nephew, these factors include: risks related to the impact of Covid, such as the depth and longevity of its impact, government actions and other restrictive measures taken in response, material delays and cancellations of elective procedures, reduced procedure capacity at medical facilities, restricted access for sales representatives to medical facilities, or our ability to execute business continuity plans as a result of Covid; economic and financial conditions in the markets we serve, especially those affecting healthcare providers, payers and customers (including, without limitation, as a result of Covid); price levels for established and innovative medical devices; developments in medical technology; regulatory approvals, reimbursement decisions or other government actions; product defects or recalls or other problems with quality management systems or failure to comply with related regulations; litigation relating to patent or other claims; legal and financial compliance risks and related investigative, remedial or enforcement actions; disruption to our supply chain or operations or those of our suppliers (including, without limitation, as a result of Covid); competition for qualified personnel; strategic actions, including acquisitions and dispositions, our success in performing due diligence, valuing and integrating acquired businesses; disruption that may result from transactions or other changes we make in our business plans or organisation to adapt to market developments; relationships with healthcare professionals; reliance on information technology and cybersecurity; disruptions due to natural disasters, weather and climate change related events; changes in customer and other stakeholder sustainability expectations; changes in taxation regulations; effects of foreign exchange volatility; and numerous other matters that affect us or our markets, including those of a political, economic, business, competitive or reputational nature. Please refer to the documents that Smith+Nephew has filed with the U.S. Securities and Exchange Commission under the U.S. Securities Exchange Act of 1934, as amended, including Smith+Nephew's most recent annual report on Form 20-F, which is available on the SEC’s website at www. sec.gov, for a discussion of certain of these factors. Any forward-looking statement is based on information available to Smith+Nephew as of the date of the statement. All written or oral forward-looking statements attributable to Smith+Nephew are qualified by this caution. Smith+Nephew does not undertake any obligation to update or revise any forward-looking statement to reflect any change in circumstances or in Smith+Nephew's expectations.
◊ Trademark of Smith+Nephew. Certain marks registered in US Patent and Trademark Office.





